Article Series
TL;DR — What Is Subcritical Water Hydrolysis?
Subcritical Water Hydrolysis (SWH) is a zero-emission treatment process that uses pressurised, high-temperature water (100°C–374°C) to act as a powerful solvent, breaking down complex organic waste into reusable materials — without combustion or added chemicals. This is the core technology inside the PHANTOM waste treatment machine — purpose-built to bring subcritical water hydrolysis to industrial scale.
At the molecular level, this hydrothermal process works by fundamentally changing the properties of water:
- The "Triple Point" Science: Between 100°C and 374°C under high pressure, water's ionic product increases 1,000-fold, allowing it to act simultaneously as an acid and a base.
- Rapid Decomposition: The ion-rich water cleaves the bonds of proteins, lipids, and plastics in just 30–50 minutes.
- Zero Dioxins: Operating in a sealed, oxygen-free liquid environment makes the combustion reactions that create toxic dioxins and furans thermodynamically impossible.
- Reusable Output: The process reduces waste volume by ~60% and sterilises pathogens (>99.9%), converting the remaining mass into liquid fertiliser, compost, or solid fuel.
Applicable to: Facilities where incineration is cost-prohibitive (high-moisture waste), chemically restricted (EU IED 2024 dioxin limits), or carbon-penalised (EU/UK ETS from 2028) — including food processors, agricultural operations, hospitals, and industrial manufacturers processing mixed organics and plastics.
Example: Instead of burning fish offal in an incinerator requiring $150+/tonne in diesel to sustain combustion on high-moisture waste, SWH uses the existing water content — heated to 250°C under pressure — to produce a sterile, sellable amino-acid fertiliser at ~$33 per 3-tonne cycle, with zero dioxin output. Run your own numbers: ROI calculator on the machine page →
Read the full scientific breakdown below for peptide bond cleavage mechanics, dielectric constant shifts, and comparisons against enzymatic hydrolysis. Related: Zero-Emission Industrial Waste Treatment Guide · Medical Waste TCO: Autoclave vs. Incineration vs. Hydrolysis

Subcritical Water Hydrolysis (SWH) is not a chemical trick. It is a precise manipulation of water's physical state — one that unlocks extraordinary reactive power without combustion, without added solvents, and without toxic byproducts. If you are evaluating non-incineration treatment for infectious medical waste, sustainable processing of fishery and slaughterhouse wet waste, or the chemical recycling of industrial plastics — the underlying science enabling all of these applications is the same: subcritical water hydrolysis. This article dismantles the physics, the chemistry, and the engineering logic from first principles.
PHANTOM ECOTECH — KNOWLEDGE SERIES
SWH Technology Series — All Guides
This article is the science foundation. Each guide below applies SWH to a specific industry, waste stream, or business decision. Find your vertical and go deep.
Medical & Healthcare
Infectious Medical Waste Non-Incineration Guide
How NHS trusts and private hospitals eliminate clinical waste incineration costs without outsourcing or autoclave bottlenecks.
Hospital PPE & Single-Use Plastics Management
Why PVC gloves, IV bags, and single-use surgical kit generate compounded dioxin risk in incinerators — and the SWH alternative.
Medical Waste TCO: Autoclave vs Incineration vs Hydrolysis
Full 5-year cost model comparing capital, operating, compliance, and disposal costs across all three treatment pathways.
UK Medical Waste Permits & IStAATT Validation
The regulatory roadmap for deploying on-site SWH in the UK: EA permits, IStAATT validation, and HTM 07-01 compliance.
Nursing Home Adult Diaper Disposal Cost Reduction
How care homes processing 200–800 kg of incontinence waste per day cut disposal costs with on-site SWH.
Agriculture & Livestock
Livestock Manure to Organic Fertilizer via SWH
Converting antibiotic-contaminated manure into pathogen-free biostimulant fertilizer: process parameters, output specs, and agronomic validation.
Fishery & Slaughterhouse Wet Waste Treatment
How fish offal, shells, blood water, and processing residues are hydrolyzed into peptide-rich compost and amino acid fertilizer within 30 minutes.
Industrial & Manufacturing
Hydrolysis for Hard-to-Recycle Industrial Plastics
PET, PE, PP, and PS depolymerization pathways, monomer recovery rates, and fuel-grade output specs for industrial plastic waste streams.
Manufacturing Waste Carbon Footprint Reduction
How SWH helps manufacturers hit Scope 3 emission targets by eliminating incineration and landfill from the waste disposal chain.
UK EPR 2025: Mixed Plastic Waste Compliance Costs
Why mixed industrial plastic costs UK manufacturers over £1,000/tonne in EPR fees, PRN costs, and Plastic Packaging Tax — and why mechanical recycling won't fix it.
ROI of an Industrial Waste Processing Machine
Payback period, tipping fee elimination, and output revenue modelling for a PHANTOM 3M3 unit at medium industrial throughput.
Foundation Guide
Zero-Emission Industrial Waste Treatment — Full Technical Guide
Process engineering, regulatory compliance, waste stream compatibility, and system specifications in one document.
What Exactly Is Subcritical Water Hydrolysis?

Subcritical Water Hydrolysis (SWH) is a zero-emission process that uses pressurised water at 100–374°C as a natural solvent to break down organic waste into reusable outputs — without combustion, added chemicals, or toxic by-products.
Three properties define what makes water subcritical:
- Dielectric constant drops from ~78.5 to ~27 — matching acetone in polarity
- Ionic product increases 1,000-fold, generating H⁺/OH⁻ at acid-catalyst concentrations
- Hydrogen-bond network loosens, enabling penetration of hydrophobic polymer chains
Applies to: Organic and plastic waste streams where high moisture content makes incineration uneconomic. Does NOT apply to glass, metal, or stone — which require pre-sorting before treatment.
Example: Fish offal at 75% moisture is unburnable. At 250°C under pressure, SWH uses the waste's own water content to hydrolyse proteins into sterile amino-acid fertiliser at ~$33/cycle — with zero dioxin output.
The word "subcritical" defines the operating window precisely. Water has a thermodynamic critical point at 374°C and 22.1 MPa. Above this point, water becomes supercritical — a gas-like phase with extreme oxidizing properties. Below this point, but well above its standard boiling threshold, water enters the subcritical state: still liquid, but radically transformed at the molecular level.
"Hydrolysis" refers to the bond-cleavage mechanism — literally water-splitting. Ester bonds, peptide bonds, glycosidic bonds, and ether linkages in organic matter are severed by the H⁺ and OH⁻ ions that subcritical water generates in high concentration. The combination — subcritical water as the hydrolysis medium — produces a reaction that is faster than enzyme digestion, cleaner than acid/base hydrolysis, and categorically safer than incineration.
The scientific basis for SWH's effectiveness was partly established through geochemical research into natural hydrothermal systems — specifically, studies of how water chemistry changes under subsurface temperature and pressure conditions drive organic matter transformation in the Earth's crust. Research into hydrogen isotope fractionation in thermally generated natural gases (Clayton, C.; Organic Geochemistry field) established that water at elevated temperature and pressure dramatically increases its ionic dissociation, producing H⁺ and OH⁻ concentrations orders of magnitude higher than ambient water. Engineered SWH systems replicate this chemistry at industrial scale — compressing what occurs over geological timescales into a 30–50 minute processing cycle.
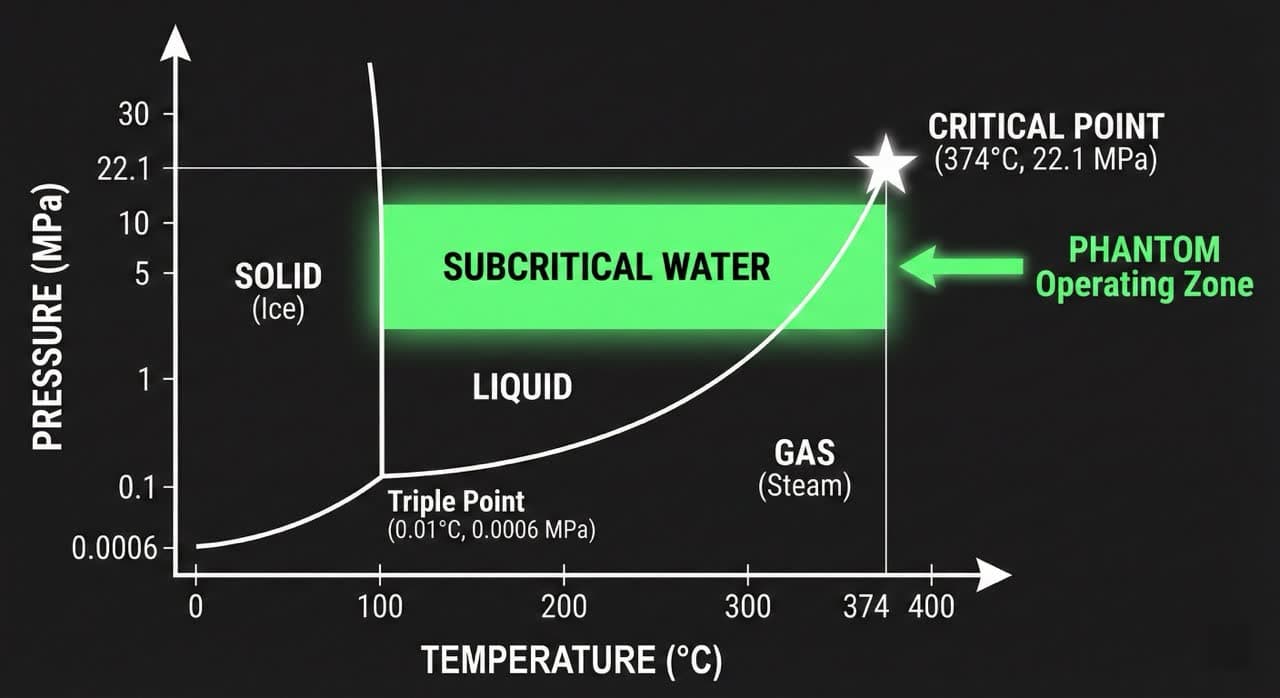
What Is the "Triple Point" Science? Why Does Water Become a Solvent Between 100°C and 374°C?

Below 374°C and above 100°C — under applied pressure — water remains liquid but undergoes a fundamental shift in its dielectric constant, ionic product, and hydrogen-bond density. These changes transform it from a polar solvent into a near-universal organic solvent capable of hydrolyzing complex polymers without any added reagent.
At ambient conditions (25°C, 0.1 MPa), liquid water has a dielectric constant (ε) of approximately 78.5 and an ionic product (Kw) of 10⁻¹⁴. These values make ambient water excellent at dissolving salts and polar molecules — but poor at attacking the covalent bonds of organic polymers (proteins, fats, cellulose, plastics).
Raise the temperature to 250°C under 5–10 MPa of pressure. Now the dielectric constant drops toward ~27 — comparable to acetone or methanol — while the ionic product (Kw) rises to approximately 10⁻¹¹: a 1,000-fold increase in H⁺ and OH⁻ ion concentration versus ambient water.
This is not a gradual change. It is a step-change in reactivity.
The weakening of water's hydrogen-bond network at elevated temperature reduces structural clustering of water molecules. Individual H₂O molecules become more mobile, more reactive, and more capable of penetrating the hydrophobic regions of organic polymers. The elevated ionic concentration means H⁺ and OH⁻ are available at concentrations sufficient to catalyze hydrolysis at the reaction rate of a strong acid or base — but with no acid or base added to the system.
The pressure's role is purely physical: it suppresses vaporization, keeping water in the liquid phase above 100°C and preserving the dense, ion-rich medium required for hydrolysis. Without pressure, water would flash to steam and reaction efficiency would collapse.
The key insight: temperature drives the chemistry; pressure preserves the medium.
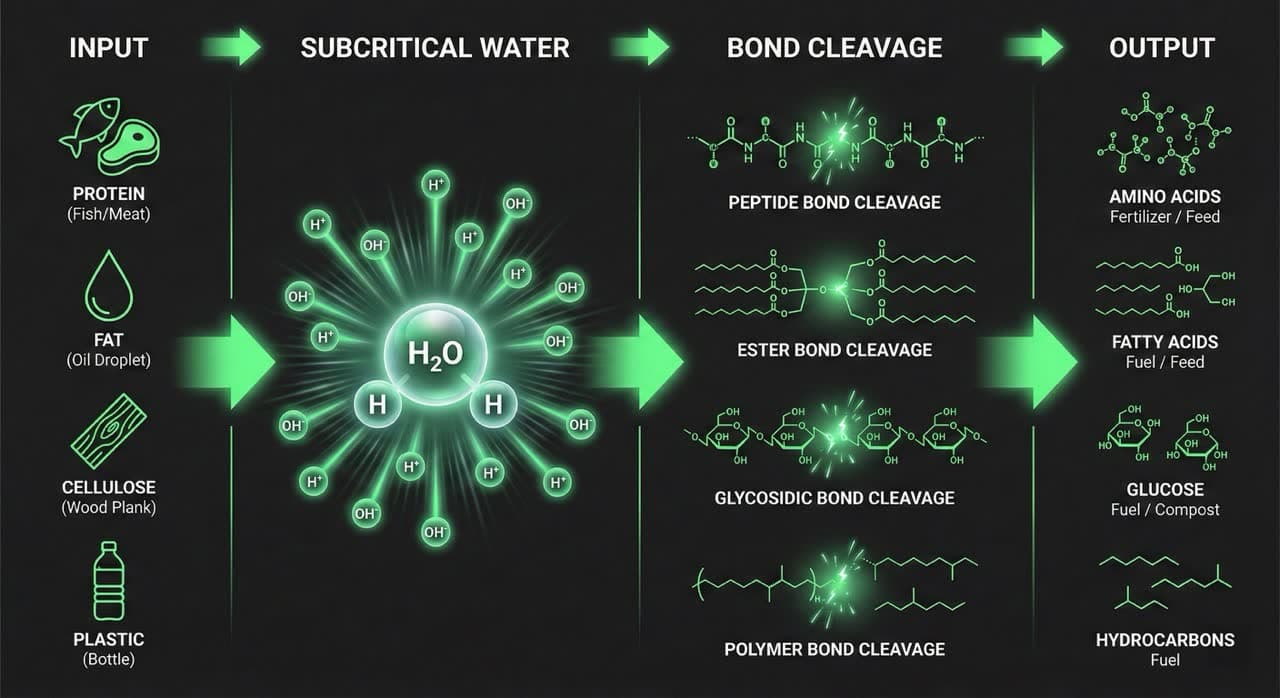
The Reaction: How Do Complex Polymers Break Down Into Simple Molecules?
In subcritical water, long-chain organic polymers — proteins, lipids, cellulose, and synthetic polymers — undergo rapid chain scission into their constituent monomers: amino acids, fatty acids, simple sugars, and short hydrocarbons. The mechanism is direct ionic catalysis, not combustion.
Proteins (Peptide Bond Hydrolysis)
Proteins are polypeptide chains — amino acids linked by peptide (–CO–NH–) bonds. Elevated H⁺ concentration attacks the carbonyl carbon of each peptide bond:
–CO–NH– + H₂O → –COOH + H₂N–
(peptide bond) (carboxylic acid) + (amine)
The products are free amino acids — directly usable as livestock feed supplements, soil fertilizers, or fermentation substrates. The PHANTOM system processes organic waste from fisheries and livestock manure through exactly this pathway. For the full treatment economics applied to livestock manure conversion to organic fertilizer via subcritical hydrolysis, see the dedicated case study.
Lipids (Ester Bond Hydrolysis)
Triglycerides contain ester bonds (–COO–) that undergo rapid hydrolysis at subcritical temperatures:
Triglyceride + 3H₂O → Glycerol + 3 Fatty Acids
The resulting fatty acids are high in calorific value. When the input stream is loaded with lipid-rich waste (food waste, fish offal, slaughterhouse residues), the output contains solid material with an energy density approaching ~5,000 kcal/kg — usable as high-calorie solid fuel. For wet waste streams, see the full analysis of fishery and slaughterhouse wet waste treatment.
Cellulose and Lignocellulosic Materials (Glycosidic Bond Hydrolysis)
Cellulose is a β-1,4-glycosidic chain of glucose units — among the most stable bonds in biological systems, notoriously resistant to enzymatic attack without pretreatment. In subcritical water above 200°C, glycosidic bonds hydrolyze rapidly:
(C₆H₁₀O₅)ₙ + nH₂O → nC₆H₁₂O₆
(cellulose) (glucose)
For paper waste, wooden building materials, and fabric inputs, this pathway generates fermentable sugars and compostable biomass.
Synthetic Polymers (Ether and Ester Bond Scission)
Plastics such as PET, PE, PP, and PS contain ester or ether linkages accessible to SWH above 250°C. PET depolymerizes to terephthalic acid and ethylene glycol. The PHANTOM system classifies processed plastic output as "reducible" fuel feedstock — volume is significantly reduced, and hydrocarbon fragments retain energy value. This chemical accessibility is the foundation of subcritical water hydrolysis for industrial plastics, which allows for the recovery of valuable monomers and volume reduction without combustion. For UK producers facing packaging levy obligations under Extended Producer Responsibility, see our UK EPR packaging compliance for plastic waste cost guide.
Net result across all organic streams: ~60% volume reduction, >99.9% pathogen kill at temperatures above 150°C, and conversion of heterogeneous waste into categorized, reusable outputs.

Why Is SWH Safer Than Incineration? The Dioxin, Furan, and NOₓ Problem Explained

Incineration generates dioxins, furans, NOₓ, particulate matter, and heavy metal-laden ash — all byproducts of incomplete combustion at atmospheric oxygen conditions. Subcritical water hydrolysis operates in a sealed, oxygen-free vessel at sub-combustion temperatures. No combustion means no combustion byproducts.
This is thermochemical first principles, not a regulatory preference.
The Dioxin Formation Pathway in Incinerators
Polychlorinated dibenzo-p-dioxins (PCDDs) and polychlorinated dibenzofurans (PCDFs) form in incinerators through two mechanisms:
- De novo synthesis — Carbon, hydrogen, oxygen, and chlorine recombine in the 200–400°C post-combustion flue gas zone, catalyzed by copper and iron particulates on fly ash surfaces.
- Precursor reactions — Chlorinated benzenes and phenols present in waste rearrange under thermal stress.
Controlling dioxin formation requires rapid quench cooling, activated carbon injection, and fabric filter systems — all adding capital and operational cost. Even with these controls, trace PCDD/PCDF emissions in incinerator flue gas remain a regulatory concern across the EU, US, and Japan.
Why SWH Has No Dioxin Formation Pathway
The PHANTOM system's sealed pressure vessel operates in a liquid-phase, oxygen-excluded environment. The conditions required for dioxin synthesis — atmospheric oxygen, gas-phase chlorinated precursors, particulate catalytic surfaces at 200–400°C — are absent by design.
| Parameter | Incineration | Subcritical Water Hydrolysis |
|---|---|---|
| Operating medium | Gas phase (air + O₂) | Liquid phase (H₂O, sealed) |
| Temperature | 850–1,200°C | 150–374°C |
| Oxygen presence | Required (combustion) | Excluded (pressurized vessel) |
| Dioxin/Furan formation | Confirmed pathway present | No formation pathway |
| NOₓ emissions | Present (thermal NOₓ) | None |
| CO₂ source | Waste combustion (direct) | Kerosene boiler only (indirect) |
| Pathogen kill mechanism | Thermal combustion | Ionic hydrolysis + steam sterilization |
| Residue | Toxic ash (landfill disposal required) | Sterile organic powder (reusable) |
| Volume reduction | ~70–90% (mass lost as CO₂) | ~60% (mass retained as product) |
For medical waste streams — where chlorinated plastics (PVC packaging, IV bags) and biohazardous material create compounded dioxin risk in incinerators — the safety differential is operationally critical. This is examined in detail in our infectious medical waste non-incineration guide and in the specific context of hospital single-use plastics and PPE waste management.

PHANTOM is the only industrial-scale system engineered to eliminate this formation pathway entirely. See PHANTOM specifications and request a quote →
How Does SWH Compare to Other Hydrolysis Methods?
Subcritical water hydrolysis is faster than enzymatic hydrolysis, cleaner than acid/base hydrolysis, and more appropriate for industrial organic waste streams than supercritical water oxidation.
| Method | Catalyst Required | Reaction Time | Hazardous Byproducts | Neutralization Step | Industrial Scale |
|---|---|---|---|---|---|
| Enzymatic Hydrolysis | Enzyme (costly, temperature-sensitive) | Hours–days | Low | No | Limited throughput |
| Acid Hydrolysis | H₂SO₄ / HCl | Minutes–hours | High (acid waste, corrosion) | Yes | High CAPEX |
| Alkaline Hydrolysis | NaOH / KOH | Hours | Moderate (caustic waste) | Yes | Regulated effluent handling |
| Supercritical Water Oxidation | None (O₂ added) | Minutes | Low — extreme pressure | No | Very high CAPEX (>22.1 MPa) |
| PHANTOM SWH | None (H₂O only) | 30–50 min | Near-zero | No | Up to 3 T/batch |
No reagent procurement. No effluent neutralization. No hazardous waste stream from the treatment system itself. Inputs: waste, water, and kerosene for the boiler. Outputs: categorized reusable materials.

Hydrothermal Treatment Methods: HTL vs SWH vs SCWO Compared
Water-based thermochemical treatment encompasses three distinct process regimes — Subcritical Water Hydrolysis (SWH), Hydrothermal Liquefaction (HTL), and Supercritical Water Oxidation (SCWO) — each defined by temperature and pressure relative to water's critical point (374°C, 220 bar). Understanding the differences determines which technology is appropriate for a given feedstock and target output.
Water-based thermochemical treatment of organic matter encompasses three distinct process regimes, each defined by temperature and pressure relative to water's critical point (374°C, 220 bar).
The Three Regimes
| Parameter | Subcritical Water Hydrolysis (SWH) | Hydrothermal Liquefaction (HTL) | Supercritical Water Oxidation (SCWO) |
|---|---|---|---|
| Temperature range | 150–374°C | 250–374°C | >374°C |
| Pressure | 5–22 MPa (subcritical) | 10–25 MPa (subcritical to near-critical) | >22 MPa (supercritical) |
| Water state | Liquid (pressurised) | Liquid to near-critical | Supercritical fluid |
| Primary reaction | Hydrolysis — bond cleavage via H⁺/OH⁻ | Depolymerisation → bio-crude oil | Oxidative mineralisation |
| Primary output | Sterile organic residue + liquid concentrate | Bio-crude oil (35–55% yield) | CO₂ + H₂O + mineral ash |
| Oxygen requirement | None | None | Required (oxidant added) |
| Carbon fate | Preserved in solid/liquid residue | Converted to bio-oil | Fully oxidised to CO₂ |
| Best feedstock | Mixed organic waste, medical waste, food waste, plastics | Wet algae, sewage sludge, lignocellulosic biomass | Highly toxic aqueous waste streams |
| Primary purpose | Waste sterilisation, volume reduction, resource recovery | Biofuel production | Destruction of toxic compounds |
| CO₂ emissions | Near-zero from vessel (boiler only) | Low (no combustion in vessel) | CO₂ is the primary carbon output |
| Scalability | Industrial (0.5–3 T/cycle, batch) | Laboratory to pilot scale (largely) | Industrial (niche — toxic waste) |
Key Distinction: HTL Converts Carbon to Fuel; SWH Preserves It for Reuse
The most important distinction between HTL and SWH is their intent and carbon pathway.
Hydrothermal liquefaction is fundamentally a biofuel production technology. Its target output is bio-crude oil — a fuel substitute derived from the conversion of biomass carbon into hydrophobic hydrocarbon compounds. The process operates in the high-temperature subcritical to near-critical regime where lipids, proteins, and carbohydrates depolymerise into oil-phase products. Feedstock selection strongly influences yield: high-lipid materials (algae, sewage sludge) produce higher oil fractions than lignocellulosic materials.
Subcritical water hydrolysis operates in a lower-temperature subcritical regime focused on hydrolytic bond cleavage rather than depolymerisation to oil. The H⁺ and OH⁻ ions generated at subcritical conditions attack ester, peptide, glycosidic, and ether bonds, breaking macromolecules into low-molecular-weight water-soluble and solid components. The carbon is not converted to oil — it remains in organic form as a sterilised residue suitable for composting, fertiliser, or solid fuel.
This makes SWH the appropriate technology when the goal is:
- Sterilisation and detoxification (medical waste, infectious organic matter)
- Volume reduction with resource recovery (food waste, livestock manure, seafood processing waste)
- Hazardous compound destruction without combustion (PCB-containing materials, dioxin precursors)
And HTL the appropriate technology when the goal is:
- Biofuel production from high-lipid wet biomass
- Energy recovery from sewage sludge or algae
The Geochemical Connection: Natural Hydrothermal Processes
Both HTL and SWH are inspired by geochemical processes occurring naturally in the Earth's crust. Deep subsurface water, heated by geothermal gradients and confined under lithostatic pressure, drives hydrolytic and thermolytic reactions in organic-rich sediments — the same chemistry responsible for petroleum formation over geological time.
Research into hydrogen isotope systematics of thermally generated natural gases established foundational understanding of how water chemistry changes under high temperature and pressure, informing the development of engineered hydrothermal treatment systems. The ionic product of water (Kw) increases by several orders of magnitude between ambient conditions and 250°C, dramatically increasing hydronium and hydroxide concentrations and explaining the accelerated reaction rates observed in both geological and engineered hydrothermal contexts.
This geochemical literature is the scientific basis from which industrial hydrothermal treatment technologies — including HTL, SWH, and hydrothermal carbonisation (HTC) — were derived.
Relationship to Hydrothermal Carbonisation (HTC)
A fourth water-based process, hydrothermal carbonisation (HTC), operates at lower temperatures (180–250°C) and produces hydrochar — a carbon-rich solid with soil amendment or fuel applications. HTC is particularly suited to high-moisture feedstocks where drying for conventional pyrolysis would be energy-prohibitive.
The four water-based thermal processes form a continuum:
HTC (180–250°C) → SWH (150–374°C) → HTL (250–374°C) → SCWO (>374°C)
Hydrochar output → Sterile residue → Bio-crude oil → Full mineralisation
Each occupies a distinct niche based on feedstock characteristics, target output, and operational constraints. For PHANTOM's positioning within this continuum — and how it compares against incineration and composting — see the PHANTOM system comparison.
Sources for this section: Peterson et al. (2008) "Thermochemical biofuel production in hydrothermal media" — Energy & Environmental Science · Toor et al. (2011) "An overview on catalytic upgrading of bio-oils" — Energy · Brunner (2009) "Near and supercritical water" — Journal of Supercritical Fluids · Möller et al. (2011) "Hydrothermal processes in nature and technology" · Savage (1999) "Organic chemical reactions in supercritical water" — Chemical Reviews.
What Are the Industrial Applications of Subcritical Water Hydrolysis?

SWH is applicable to any organic waste stream where hydrolyzable bonds are present — spanning livestock waste, fishery residues, medical waste, food processing waste, and plastic fractions.
Livestock and agricultural waste — Manure and urine are processed into compost and liquid fertilizer. High-temperature sterilization eliminates antibiotic residues and pathogens that survive conventional composting. Liquid output diluted 500× with seawater produces a biostimulant fertilizer. Read the full breakdown of converting livestock manure to organic fertilizer via subcritical hydrolysis.
Fishery and aquaculture residues — Fish offal, shells, and processing waste are hydrolyzed to produce peptide-rich compost and amino acid-concentrated liquid fertilizer. Collagen and keratin fractions — resistant to ambient biodegradation — are fully solubilized within 30 minutes. For a deeper analysis, refer to the case study on fishery and slaughterhouse wet waste treatment.
Medical and infectious waste — PPE, diapers, biological waste in plastic containers, and sharps packaging are sterilized (>99.9%) and volume-reduced by ~60%. Output is classified as non-infectious solid material, eliminating the biohazard logistics chain. Glass, metal, and stone require pre-sorting. Regulatory requirements for UK on-site treatment are covered in detail in our UK medical waste permits and validation guide.
Plastic and fuel-stream waste — PET, PE, PP, PS, wooden materials, rubber, and paint yield high-calorie solid fuel (~5,000 kcal/kg). This has significant manufacturing waste and carbon footprint reduction implications, helping facilities meet increasingly stringent Scope 3 emission targets.
For full throughput specifications across all three PHANTOM models (0.5t, 1t, 3t), including utility requirements and ROI modelling, see the PHANTOM organic waste treatment machine buyer's guide.
Technical Specifications
Phantom Ecotech — Technical Reference
Subcritical Water Hydrolysis: Key Parameters
Ionic Reactivity vs. Ambient Water (K𝑤)
SWH vs. Incineration vs. Landfill
| Parameter | Incineration | Landfill | PHANTOM SWH |
|---|---|---|---|
| Dioxin emissions | Present | Leachate risk | None |
| Pathogen kill rate | High (combustion) | Low | >99.9% |
| Output usability | Toxic ash (disposal) | None | Compost / Fuel / Feed |
| Chemical additives | None (fuel) | None | None (H₂O only) |
| CO₂ emission source | Waste combustion | CH₄ + CO₂ off-gas | Boiler only |
| Ionic reactivity of medium | N/A | N/A | Kw ~10⁻¹¹ (×1,000) |
Ready to specify SWH for your facility?
Input stream analysis · Throughput sizing · Site assessment
What Are the Current Technical Limitations of SWH?

High initial CAPEX for pressure vessel fabrication, inorganic fraction exclusion, and per-cycle energy input are the primary engineering constraints. They are quantifiable and manageable — not prohibitive.
The constraints stated plainly:
- Pressure vessel materials — The SUS 304 stainless steel reactor requires monitoring for corrosion under high-chloride inputs (fishery waste, certain medical streams). Gasket and seal replacement is specified at ~10-year intervals under normal operation.
- Inorganic exclusion — Glass, metal, and stone must be pre-sorted. They will not hydrolyze and remain as inert residue. Effective daily throughput must account for pre-sort time.
- Energy input — The kerosene boiler represents the primary operational cost. Benchmark this against landfill tipping fees, incineration gate fees, and transport costs for your specific waste volumes.
- Throughput ceiling — The Phantom 3M3 model processes 3 tonnes per input cycle with a 30-minute reaction time. Effective daily throughput at 20–22 operational hours is ~36–44 tonnes/day. Facilities requiring higher throughput require parallel units.
Total cost of ownership, benchmarked against landfill tipping fees and incineration gate fees for your specific waste volume, is the correct calculation. A full financial comparison of on-site treatment options is available in our medical waste TCO analysis: autoclave vs. incineration vs. hydrolysis.
Conclusion: From "What Is It?" to "Why Does It Matter?"
Subcritical water hydrolysis is water doing chemistry that acid, enzymes, and fire cannot do simultaneously — at industrial throughput, without toxic byproducts, with a reusable output stream. The physics are proven. The engineering is commercially deployed. The regulatory trajectory — EU Green Deal targets, rising landfill taxes, tightening dioxin emission limits across global markets — is moving in one direction.
The remaining question is not scientific. It is operational: when does your facility make the transition? For the full regulatory and financial picture — covering how landfill tax, EPR fees, PRN costs, and CBAM exposure stack for UK manufacturers — see: manufacturing waste reduction UK: the complete compliance guide. For facilities processing difficult industrial plastic streams, the engineering decision starts with input classification — see the Industrial Plastics Subcritical Hydrolysis Specification Guide for polymer compatibility, Track A vs Track B separation, and PCB vessel requirements.
⚙️ Tanaka: Stop modelling the chemistry. The hydrolysis reaction is optimized. What requires your engineering attention now is input characterization — specifically moisture content, chloride concentration, and inorganic fraction percentage of your specific waste stream. These three parameters determine your pre-sorting requirement, boiler fuel consumption, and output classification. Bring those numbers to the first conversation.
Frequently Asked Questions
Subcritical water is liquid water maintained between 100°C and 374°C under sufficient pressure to prevent vaporization. Supercritical water exceeds both the critical temperature (374°C) and critical pressure (22.1 MPa), entering a gas-like state with extreme oxidizing power. SWH targets hydrolysis into reusable resource fractions; supercritical water oxidation (SCWO) targets complete mineralization. PHANTOM operates subcritically — the optimal zone for resource recovery rather than total destruction.
No. The reaction medium is water only. No acids, bases, enzymes, or oxidizing agents are added. This eliminates reagent procurement cost, effluent neutralization steps, and chemical handling risk entirely.
Yes, with the constraint that glass, metal, and stone must be pre-sorted. Mixed organic streams can be co-processed. Selective single-stream batches produce higher-purity, higher-value outputs — for example, amino acid fertilizer from pure fish waste versus general compost from mixed organics.
ASME Boiler & Pressure Vessel Code, PED (EU Pressure Equipment Directive), CE marking, and KS (Korean Standards). Documentation is available on request via our contact page.
The primary maintenance requirement is periodic inspection and gasket/packing replacement approximately every 10 years. Operating costs consist primarily of kerosene fuel for the boiler. No complex chemical treatment systems require servicing. Designed service life exceeds 10 years under standard operating cycles.
Part of Phantom Ecotech's Zero-Emission Industrial Waste Treatment knowledge series. Full system specifications, ROI calculator, and facility-type comparisons: PHANTOM Waste Treatment Machine →
Cite this article: Phantom Ecotech Research Team. (2026, March). "What Is Subcritical Water Hydrolysis? — Including HTL vs SWH vs SCWO Comparison." Phantom Ecotech. https://phantomecotech.com/blog/what-is-subcritical-water-hydrolysis#htl-swh-comparison
Published: March 2026. Last updated: March 2026. Author: Phantom Ecotech Research Team.
Sources: National Institute of Standards and Technology (NIST) Thermophysical Properties of Fluid Systems; Brunner, G. (2009). Near and supercritical water. Part I: Hydrothermal processes. Journal of Supercritical Fluids. IStAATT International Standards (2011). Criteria for Treatment Technologies. NHS England HTM 07-01 (2023). Environment Agency, Healthcare waste: appropriate measures for permitted facilities (December 2021). DEFRA Digital Waste Tracking policy paper (February 2026). Peterson et al. (2008). Thermochemical biofuel production in hydrothermal media. Energy & Environmental Science. Toor et al. (2011). An overview on catalytic upgrading of bio-oils. Energy. Savage, P.E. (1999). Organic chemical reactions in supercritical water. Chemical Reviews.
⚠️ Disclaimer: The technical data, process parameters, and performance figures in this article are compiled from published scientific literature, regulatory guidance, and manufacturer specifications, and are provided for general informational and educational purposes only. They do not constitute engineering, legal, financial, or procurement advice. System performance varies by input stream composition, operating conditions, and site-specific factors. Always conduct independent technical due diligence before specifying or procuring any waste treatment equipment. Phantom Ecotech accepts no liability for decisions made in reliance on this article without independent professional and engineering verification.



