Article Series
Getting approval for on-site clinical waste treatment in the UK takes between 12 and 24 months and costs £20,000–£50,000+ before you treat a single bag. If you have already read our guide to UK on-site medical waste treatment permits and validation, you know that a bespoke Environment Agency permit is the only route — no exemption, no shortcut. This article gives you the exact stage-by-stage timeline, the costs at each step, and the specific failure points that kill most first-time applications before the EA ever reviews them.
Why you may see higher figures elsewhere: The 12–24 month and £20,000–£50,000+ figures here reflect the general EA permitting process for all alternative treatment technologies — including autoclaves with existing type-approval data, where private lab testing costs are lower (£5,000–£15,000). If you are deploying a novel technology with no prior UK type-approval — such as subcritical water hydrolysis — expect the upper end of the timeline and significantly higher costs: private microbiological lab testing alone runs £15,000–£40,000, pushing total costs to £38,000–£95,000. See the IStAATT Level III guide for novel technologies for the SWH-specific cost breakdown and failure analysis.

What Is IStAATT and Why Does the EA Require It for On-Site Clinical Waste Treatment?
IStAATT validation is the mandatory microbial kill-rate standard that every on-site clinical waste treatment system must pass before the Environment Agency will allow it to operate commercially in England. IStAATT stands for the International Society on Analytical Assessment of Treatment Technologies — an independent body whose microbial inactivation levels the EA has formally adopted in its healthcare waste appropriate measures guidance.
The four validation levels are:
- Level I — ≥6 log₁₀ reduction of vegetative bacteria, fungi, and lipophilic viruses (99.9999% kill)
- Level II — Level I plus ≥6 log₁₀ reduction of parasites, mycobacteria, and hydrophilic viruses
- Level III — Level II plus ≥4 log₁₀ reduction of Geobacillus stearothermophilus and Bacillus atrophaeus bacterial spores
- Level IV — Level III but raising the spore reduction requirement to ≥6 log₁₀ (true sterilisation)
Level III is the minimum standard required for treatment of infectious clinical waste under EWC code 18 01 03*. Level IV applies only in exceptional biohazard scenarios.
This applies when your facility treats orange-bag infectious clinical waste on-site using any alternative treatment technology — autoclave, microwave, subcritical water hydrolysis, or similar — and requires an EA environmental permit to do so. It does NOT apply to laboratory microbiological waste treated under RPS 233 (autoclave only, ≤1 tonne/day), offensive/hygiene waste, or standard industrial waste streams.
Scenario: A 180-bed NHS-contracted private hospital in the Midlands generates approximately 400 kg of infectious clinical waste per week. To treat it on-site, they need an EA bespoke permit with Level III IStAATT validation. Autoclaving at a centralised contractor facility costs them £185/tonne. The business case for on-site treatment is clear — but the route to approval is not.
What Is the Step-by-Step IStAATT Application and EA Permitting Process?
The approval pathway runs through six sequential phases. Skipping or compressing any stage delays the entire process. The EA does not accept a treatment system into operation until it has issued written approval of the validation report.
The six phases are:
- EA pre-application advice — confirm permit type, scope, and evidence format requirements
- Application preparation — assemble the full bespoke permit package including all management plans
- EA submission and duly-made check — formal validation before assessment begins
- EA determination — formal review, public consultation, technical assessment
- IStAATT Level III validation testing — three-cycle worst-case challenge test programme
- EA approval of validation report — written sign-off before operations begin
This applies to all on-site clinical waste treatment installations in England requiring a bespoke permit. It does NOT apply to Scotland (SEPA regulates), Wales (NRW), or Northern Ireland (NIEA) — those regulators have parallel but distinct processes.
Scenario: A 300-bed NHS Foundation Trust installs a subcritical water hydrolysis unit and begins Phase 1 (pre-application advice) in January 2026. Following this timeline correctly, it should be treating waste commercially by Q1–Q2 2027.
MEDICAL — UK COMPLIANCE
IStAATT Validation & EA Permit: Stage-by-Stage Timeline
England only · Bespoke permit route · Infectious clinical waste (EWC 18 01 03*) · All figures indicative
EA Pre-Application Advice
2–8 weeks
Basic: Free Enhanced: £500–£2,000+ (£100/hr + VAT)
⚠ Skipping this is the #1 cause of application failure
Application Preparation
8–24 weeks
Consultancy: £5,000–£20,000+ Internal resource: variable
⚠ Incomplete packages returned — up to £1,613 forfeited
EA Submission & Duly Made
4–6 weeks
EA permit fee: £8,000–£12,000+ + management plan fees: £1,241–£1,246 each
⚠ Each rejected resubmission = 6 week delay
EA Determination
13–26 weeks
Included in permit fee Info notices: £1,290 each (3rd+)
⚠ Novel technologies routinely exceed 13-week target
IStAATT Level III Validation Testing
4–8 weeks
Testing total: £5,000–£15,000+ (lab, BIs, microbiologist, loggers)
⚠ Wrong BI organism or non-UKAS lab = full retest required
EA Validation Report Approval
4–8 weeks
Included in permit Re-validation every 4 years: £5,000–£15,000
⚠ Operations cannot begin until written EA approval received
Indicative Total Timeline
12–24 months
Indicative Total Cost
£20,000–£50,000+
Annual EA Subsistence
£3,200–£4,800 / yr
Fee Uplift (from Apr 2026)
+3.8% CPI
Figures are indicative. Costs vary by technology complexity, application quality, and EA regional office. EA fee schedule updated annually. PHANTOM documentation packages are pre-assembled for UK submission.

What Does EA Pre-Application Advice Actually Involve — and Why Is It Non-Negotiable?
EA pre-application advice is the most important step in the entire process. It is not optional bureaucracy. For novel technologies, it is the single mechanism by which you align your evidence package format with what the EA's technical assessors will actually accept — before you spend £15,000–£40,000 on validation testing.
Pre-application advice comes in two tiers:
- Basic pre-application advice — free. Submit a general enquiry through the EA Permitting and Support Centre. Response within 20 working days. Confirms whether a bespoke permit is required, which standard rules (if any) apply, and the broad scope of your application.
- Enhanced pre-application advice — charged at £100/hour + VAT. Typically 5–10 hours (£500–£1,000+). Covers technical details specific to your treatment process: time/temperature/pressure parameters, waste stream EWC codes, challenge load design for validation testing, and evidence format requirements.
For subcritical water hydrolysis or any non-autoclave alternative treatment technology, enhanced pre-application advice is mandatory in practice — the EA explicitly offers and recommends a staged application arrangement for complex or novel technologies, where information is submitted progressively rather than all at once. Request this arrangement at the enhanced advice stage.
This applies when your technology is not a standard porous-load autoclave (the only alternative treatment technology with well-established EA precedent). It does NOT replace the formal permit application — it is pre-application only, not a decision.
Scenario: A care home group piloting a microwave treatment unit skips enhanced pre-application advice to save time. They submit a full application in April. The EA returns it in June as "not duly made" — incomplete risk assessment. The group forfeits up to £1,613 of the application fee and loses 6 weeks. Their corrected resubmission is accepted in August, resetting the determination clock.
How Much Does an EA Bespoke Permit Application Cost for Clinical Waste Treatment?
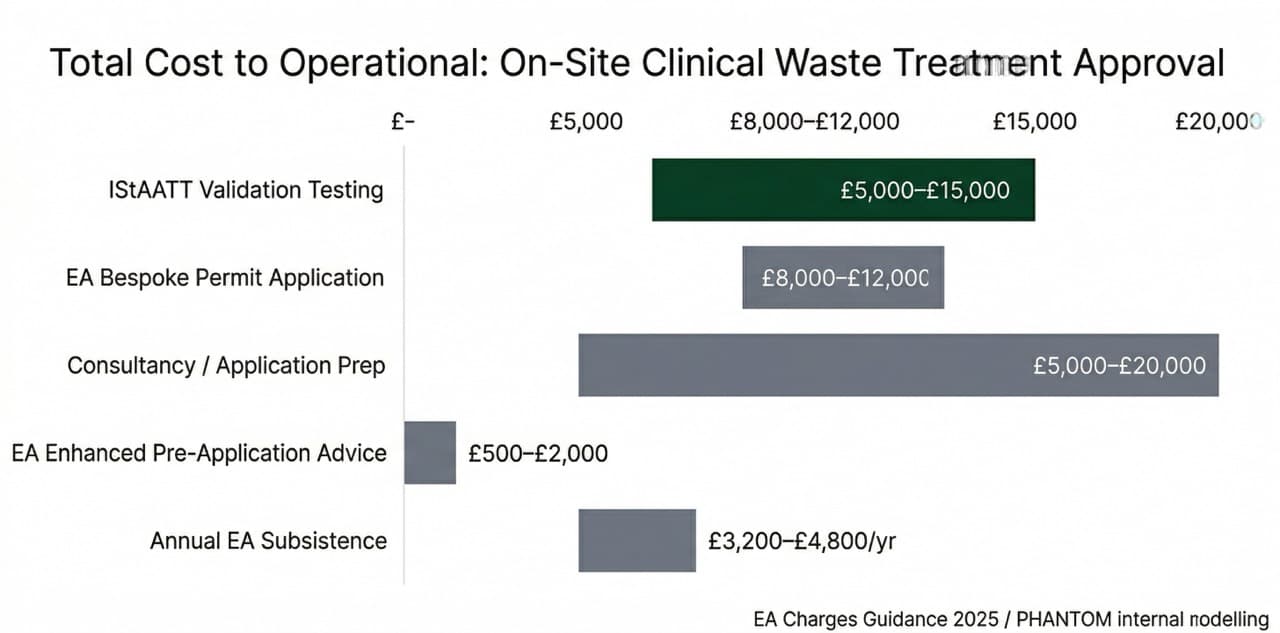
The EA application fee for a bespoke clinical waste treatment permit typically falls between £8,000 and £12,000+, depending on the complexity of the activities and the number of management plan assessments required. This is the single largest upfront cost before validation testing.
The fee structure breaks down as follows:
- Core bespoke application charge — £8,000–£12,000+ (activity-dependent)
- Odour management plan assessment — £1,241–£1,246 (charged separately)
- Emissions management plan assessment — £1,241–£1,246 (if applicable)
- Fire prevention plan assessment — £1,241–£1,246 (if applicable)
- Third or subsequent information notices — £1,290 each (if the EA requests further information on an issue it has already raised twice)
- Returned incomplete application fee penalty — up to 20% of fee retained, capped at £1,613
- Annual EA subsistence charge — approximately £3,200–£4,800/year once permit is issued (roughly 40% of the application fee)
A 3.8% CPI uplift to all EA fees takes effect 1 April 2026.
This applies to bespoke permits for on-site treatment of infectious clinical waste in England. It does NOT apply to standard rules permit applications (which are currently unavailable for clinical waste treatment) or to waste exemptions.
Scenario: A private hospital submits a bespoke application in March 2026 — just before the April fee increase. Application fee: £9,800. Odour management plan: £1,243. Fire prevention plan: £1,243. Total EA fees: £12,286. Consultancy preparation cost: £12,000. IStAATT validation testing: £9,500. Total spend before first bag is treated: £33,786.

What Does IStAATT Level III Validation Testing Actually Require?
IStAATT Level III validation requires three separate treatment cycles using a worst-case challenge load, with results analysed by a UKAS-accredited laboratory under supervision of a qualified independent microbiologist. A 4 log₁₀ reduction of bacterial spores must be demonstrated at the upper 95% confidence interval.
The specific technical requirements are:
- Biological indicators: Certified spore strips containing ≥1 × 10⁶ spores of Geobacillus stearothermophilus (wet heat/subcritical water hydrolysis processes) or Bacillus atrophaeus (chemical/dry heat) with a certified D-value ≥1.8 minutes at 121°C
- Spore placement: 3–12 strips per treatment cycle depending on plant capacity, positioned at worst-case heat penetration locations
- Control strips: Minimum 6 untreated control strips per test series
- Challenge load: Maximum batch size, including sealed rigid bins, suction canisters, fluids — minimum 5% heavy organic load (e.g., blood) by weight
- Cycle count: Three complete treatment cycles, each meeting required time/temperature/pressure parameters confirmed by thermal data loggers
- Laboratory: UKAS-accredited microbiological testing facility
- Statistical pass criterion: Log₁₀(X_T + 1.96σ) must be ≤ log₁₀(X_C) − 4 (i.e., 4 log₁₀ reduction confirmed at the upper 95% confidence interval)
- Re-validation: Required every 4 years or after any process parameter changes
This applies to all alternative treatment technologies seeking Level III clearance for infectious clinical waste. It does NOT apply to offensive/hygiene waste (EWC 18 01 04, 20 01 99) which does not require treatment validation.
Scenario: A subcritical water hydrolysis unit operating at 180°C and 10 bar conducts its three-cycle validation using G. stearothermophilus strips placed inside sealed orange bins and suction canisters at the vessel's coolest zones. UKAS laboratory analysis confirms 4.3 log₁₀ reduction at the 95% confidence interval. Pass. The validation report is submitted to the EA within 5 working days.
After Commissioning: Why HTM 07-01 Classification Is the Next Urgent Operational Risk

Once your system is validated and the EA has issued written approval, the permit is live. But the economics of on-site treatment collapse the moment waste arrives misclassified.
A system validated for orange-bag infectious waste (EWC 18 01 03*) cannot legally treat tiger-stripe offensive waste or yellow-lidded sharps containers under the same permit conditions. If wards are routinely overfilling orange bags with non-infectious waste — a widespread problem in mixed-ward environments — the treatment throughput falls, the cost-per-tonne rises, and the compliance audit trail deteriorates.
Getting the HTM 07-01 waste classification right at ward level for on-site treatment facilities is not a housekeeping task. It is a prerequisite for the treatment economics to work. Misclassification is the most common operational failure in the first 12 months after a system goes live.
What Are the Most Common Reasons IStAATT Applications Stall or Fail?
Most IStAATT-linked applications in the UK do not fail because the technology cannot meet Level III — they fail because of process errors that the EA's tightened 2024 duly-made validation catches before technical assessment even begins.
The top failure points in order of frequency:
- Submitting the application without adequate EA pre-application engagement — the EA explicitly states it will return incomplete applications; since June 2024, early validation of complex applications is standard practice
- Missing or inadequate management plans — odour, emissions, fire prevention, and pest management plans are all separately assessed and must meet sector-specific appropriate measures
- Insufficient technical competence evidence — WAMITAB/CIWM qualification documentation is mandatory and routinely omitted by first-time applicants
- Incomplete site condition report — baseline environmental data missing or insufficiently detailed
- Validation testing with non-UKAS-accredited laboratory — results are not accepted by the EA regardless of data quality
- Wrong biological indicator for the process type — B. atrophaeus submitted for a wet-heat process that requires G. stearothermophilus
- Challenge load not representative of worst-case conditions — test conducted with loose waste rather than sealed bins and high-organic content loads
- Statistical analysis at mean rather than upper 95% confidence interval — a common error that passes the mean but fails the required statistical test
This applies to bespoke permit applications in England where the treatment technology is novel or not an established porous-load autoclave. It does NOT apply to RPS 233 autoclave operations for microbiological laboratory waste, which have a simpler compliance framework.
Scenario: At a 180-bed NHS-contracted hospital in the Midlands, the team had a sound technology, a strong business case, and a qualified team. Their application stalled for 8 months because the log-reduction evidence package arrived without a completed EA pre-acceptance consultation. The EA does not evaluate a validation submission that arrives without prior pre-consultation sign-off — but this requirement is buried in guidance most facilities only discover after their first submission is returned. The result: a second application fee, a second consultancy engagement, and an £18,000 sunk cost on a validation test that had to be redesigned and repeated. The entire delay was preventable with a single enhanced pre-application advice meeting.
What Is the Difference Between the T25 Exemption and a Bespoke EA Permit for Clinical Waste?
There is no T25 exemption route for clinical waste treatment. The T25 exemption covers anaerobic digestion of food and biodegradable waste at non-agricultural premises — it has no connection to infectious clinical waste or alternative treatment technologies. This is one of the most persistent misconceptions in healthcare estates management.
The actual regulatory options for on-site clinical waste treatment are:
- Bespoke environmental permit — the only current route for infectious clinical waste treatment using alternative treatment technology. Full IStAATT Level III validation required pre-operationally.
- Standard rules permit — previously available (SR2013 No 1 for up to 100t/year clinical waste). Currently unavailable for new applications. Not a viable route.
- RPS 233 — narrow regulatory position statement for autoclaving microbiological laboratory waste only (not general clinical/infectious waste) at up to 1 tonne/day. Valid only until EA review in August 2026. Does not cover subcritical water hydrolysis or any non-autoclave technology for general infectious waste.
- T25 exemption — covers food/biodegradable waste anaerobic digestion. Completely unrelated to clinical waste treatment.
This applies to all healthcare facilities in England considering on-site treatment of orange-bag infectious waste. It does NOT apply to Scotland, Wales, or Northern Ireland — each has its own regulator with a parallel but distinct process.
Scenario: An NHS estates manager assumes the T25 exemption covers their planned on-site biodigester for clinical waste. After six months of planning against this assumption, their legal team confirms there is no such exemption for clinical waste. They restart the bespoke permit process from Phase 1. A pre-application advice call with the EA in week one of their project would have prevented this.
The Root Cause Most Applications Miss
You now understand why IStAATT applications stall. The technology is rarely the issue — Level III validation is achievable for any compliant system. The issue is process sequencing.
The single most common failure pattern is submitting log-reduction evidence without first completing EA pre-acceptance consultation. The EA will not evaluate a validation submission that arrives without prior pre-consultation sign-off on the challenge load design and evidence format. This requirement is not prominently signposted in the public guidance — most facilities discover it only after their first submission is returned. The result is a 6–12 month delay, a second application fee, and often a complete redesign of the validation test programme. Total sunk cost: £15,000–£40,000.
The root cause is not the technology or the regulation. It is the format mismatch between the evidence package a facility assembles independently and the specific format the EA's technical assessors require to even begin evaluation. That is exactly the problem the PHANTOM on-site clinical waste treatment system is designed to eliminate. PHANTOM's validation documentation package is pre-assembled for UK submission — removing the primary failure point that kills most first-time applications before they are evaluated.
The full IStAATT Level III validation and EA permitting process takes 12–24 months from initial pre-application advice to written EA operational approval. IStAATT validation testing itself takes 4–8 weeks, but this occurs at Stage 5 — after the EA determination process, which takes 13–26 weeks. Novel technologies such as subcritical water hydrolysis sit at the longer end of the timeline.
The EA bespoke permit application fee is typically £8,000–£12,000+, with separate charges of £1,241–£1,246 for each required management plan assessment (odour, emissions, fire prevention). IStAATT validation testing costs a further £5,000–£15,000+. Total cost before operations begin is typically £20,000–£50,000+, plus ongoing annual EA subsistence of £3,200–£4,800.
IStAATT Level III validation is the minimum microbial inactivation standard required by the Environment Agency for on-site treatment of infectious clinical waste. It requires a 6 log₁₀ reduction of vegetative bacteria, fungi, viruses, parasites and mycobacteria, plus a 4 log₁₀ reduction of Geobacillus stearothermophilus and Bacillus atrophaeus bacterial spores. Tests must be conducted over three worst-case challenge cycles by a UKAS-accredited laboratory.
No. The T25 waste exemption covers anaerobic digestion of food and biodegradable waste at non-agricultural premises — it has no connection to infectious clinical waste or alternative treatment technologies. The only current route for on-site infectious clinical waste treatment in England is a bespoke Environment Agency environmental permit with IStAATT Level III validation.
Most applications fail due to inadequate EA pre-application engagement — particularly submitting validation evidence without first confirming the EA's required evidence format and challenge load specification. Since June 2024, the EA returns incomplete applications with up to £1,613 in fees forfeited. Other common failures: missing management plans, insufficient technical competence documentation, non-UKAS laboratory analysis, and incorrect biological indicator organisms.
Ready to Start Your IStAATT Application — Without the £15,000–£40,000 Re-Test Risk?
PHANTOM's validation documentation package is pre-assembled for UK EA submission. The log-6 lab evidence framework, pre-consultation documentation structure, and stream-specific technical assessment requirements are all included — removing the primary failure point that sends most first-time applications back to square one.
Sources: Environment Agency IStAATT Guidance; Environmental Permitting (England and Wales) Regulations 2016; EPR 5.07 Clinical Waste Sector Guidance; EA Charges Guidance 2025/26; HTM 07-01 (Department of Health); EA Waste Permitting Transformation Programme 2025.
Disclaimer: All figures are for informational purposes only and do not constitute legal, financial, regulatory, or procurement advice. Timelines and costs are indicative based on UK 2025–2026 operational experience and publicly available EA guidance. This guide covers England only. Scotland: SEPA. Wales: NRW. Northern Ireland: NIEA. Consult a qualified environmental permitting consultant before commencing any permit application.




